In 2023, malaria contaminated practically 294 million individuals and killed shut to six,00,000. Despite early victories in the combat in opposition to malaria, world progress has stalled in recent times. The parasites are adapting, changing into proof against remedies, whereas mosquitoes are surviving pesticides. It’s a shape-shifting enemy—and the outdated instruments are slipping.
India has diminished its malaria burden by over 80% between 2015 and 2023—however final 12 months, tribal districts comparable to Lawngtlai (Mizoram) and Narayanpur (Chhattisgarh) nonetheless recorded malaria charges of over 56 and 22 circumstances per 1,000 individuals, respectively as per the National Centre for Vector Borne Diseases Control —reminders that the parasite continues to thrive in a number of pockets lengthy after nationwide averages have improved.
While Africa faces largely Plasmodium falciparum, India additionally battles the relapse-prone Plasmodium vivax which might lie dormant in the liver and reawaken weeks or, even months later. In Jharkhand, blended infections account for practically 20% of circumstances (NCVBDC), complicating elimination. Even the place incidence has dropped, the parasite can persist—lurking in asymptomatic carriers (individuals with no signs) or returning months after an infection.
The seek for smarter, longer-lasting vaccines has by no means been extra pressing.

Hope with limits: RTS,S and R21
After a long time of setbacks, the first accepted malaria vaccine—RTS,S—arrived in 2021. It provided about 55% safety in the first 12 months, however efficacy waned by 18 months, requiring a fourth booster dose.
The R21/Matrix-M vaccine, developed by Oxford and the Serum Institute, confirmed as much as 77% efficacy in Phase 3 trials successful World Health Organization (WHO) approval in 2023. Fewer doses, low price, and Indian manufacturing make it particularly promising.
Still, each vaccines goal just one stage of the parasite, leaving reinfection and transmission a lingering menace.
Whole-parasite vaccines: a stronger shot on the horizon
Instead of concentrating on a single protein, like in RTS,S and R21, whole-parasite vaccines expose the immune system to the complete malaria parasite—alive, however weakened. The experimental PfSPZ vaccine mimics pure an infection utilizing radiation-weakened P. falciparum sporozoite (the parasite’s early-stage kind) delivered instantly into the bloodstream. Early research confirmed that 96% of members developed sturdy antibodies, with as much as 79% safety after the third dose.
Building on that basis, a modified model referred to as PfSPZ-LARC2, developed by Sanaria, could push efficacy even additional. The simplicity of a one-dose routine, regardless of the intravenous requirement, may make it a robust candidate for focused use in outbreak zones or amongst hard-to-reach migrant populations in India.
Unlike vaccines that concentrate on the parasite’s earlier stage, PfRH5 acts throughout the blood stage, when signs seem and the danger of extreme sickness will increase. Since RH5 is an important protein for crimson blood cell invasion that the parasite can’t simply alter, it provides cross-strain safety—a uncommon asset in malaria vaccine design. Phase 1a/b and Phase 2b trials in the UK, The Gambia, and Burkina Faso have proven promising outcomes. These vaccines may complement earlier-stage ones and could assist enhance pure immunity in individuals who’ve beforehand had malaria.
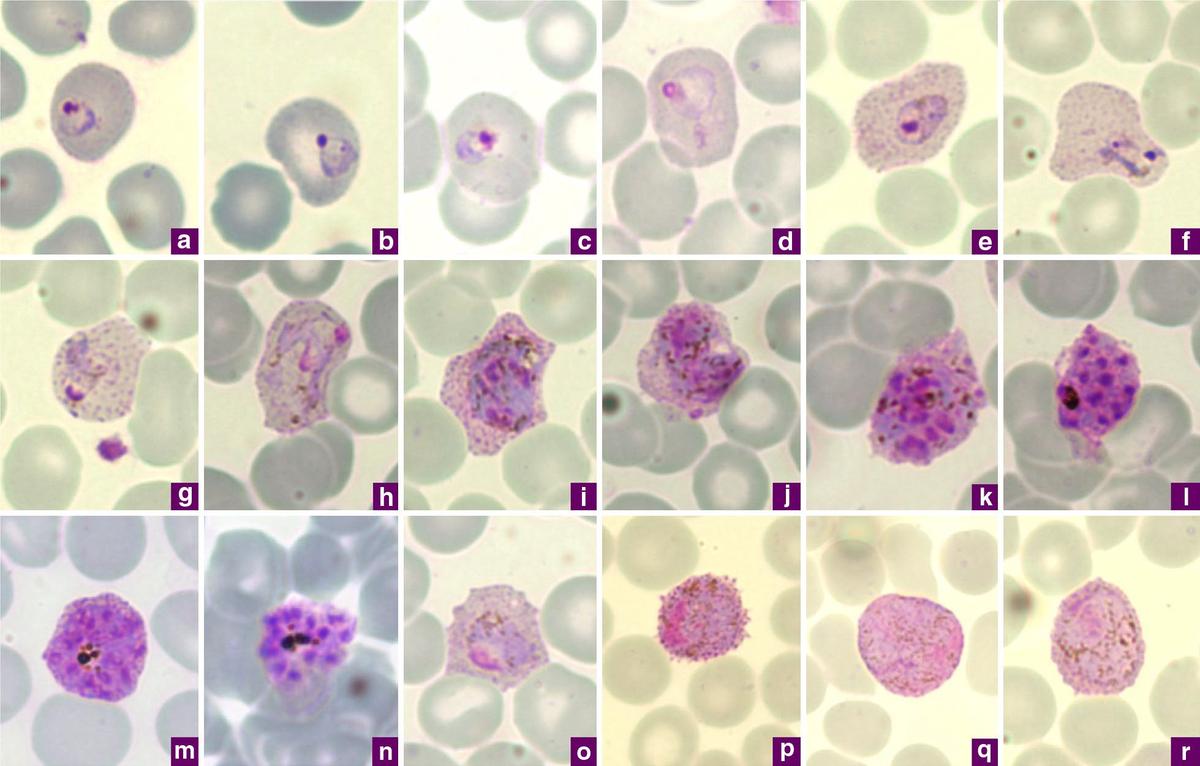
Photomicrographs of Plasmodium vivax in Giemsa-stained skinny blood movies
| Photo Credit:
By Chavatte JM, Tan SB, Snounou G, Lin RT – Chavatte JM, Tan SB, Snounou G, Lin RT (2015). “Molecular characterization of misidentified Plasmodium ovale imported cases in Singapore.”. Malar J 14: 454. DOI:10.1186/s12936-015-0985-8. PMID 26577930. PMC: 4650842.- “This article is distributed below the phrases of the Creative Commons Attribution 4.0 International License(https://creativecommons.org/licenses/by/4.0/)“, CC BY 4.0, https://commons.wikimedia.org/w/index.php?curid=122024897
Transmission-blocking vaccines
While the above vaccines goal to guard people, transmission-blocking vaccines (TBVs) goal the parasite in the mosquito—halting its unfold at the inhabitants stage. Pfs230D1 induces antibodies that forestall parasite fertilization inside the mosquito intestine. In Mali, it diminished transmission by 78% in a Phase 2 trial.
This technique is particularly related to India with a far larger proportion of asymptomatic carriers. “Our group and others in India are actively working on TBVs to address this reservoir,” stated Agam P. Singh, scientist at the National Institute of Immunology, New Delhi.
India, too, is coming into the TBV area with its personal candidates. In July 2025, AdFalciVax was introduced by the Indian Council of Medical Research (ICMR), the nation’s first indigenous dual-stage malaria vaccine. Unlike single-phase vaccines, it combines pre-erythrocytic (PfCSP) and transmission-blocking (Pfs230 and Pfs48/45) antigens to each forestall an infection and block mosquito transmission. “AdFalciVax has completed preclinical testing,” stated Subhash Singh, who leads the programme at ICMR-RMRC Bhubaneswar. In mice, it triggered sturdy immune responses lasting over 4 months—roughly equal to a decade in people—and remained steady at room temperature for 9 months, probably aiding rural deployment.
Progress can be seen past P. falciparum. A primary-in-human trial in Thailand confirmed that the P. vivax TBV Pvs230D1M diminished mosquito transmission by as much as 96%, one other ray of sunshine for India’s mixed-species numbers. India, too, shouldn’t be far behind. “A similar research program for P. vivax is underway, in collaboration with AdFalciVax co-inventors Sanghamitra Pati and Sushil Singh,” stated Dr. Singh.
Boosting immune energy
Strengthening the immune response itself is one other energetic entrance. A current protein-based vaccine mixed a ferritin nanoparticle with CpG—a kind of adjuvant, or immune booster already utilized in hepatitis B vaccines—and lower liver-stage parasite burden by 95% in mice.
AdFalciVax confirmed over 90% safety in mice even with alum, a gentle and extensively used adjuvant. “We saw protection on a par with more inflammatory adjuvants such as MPLA (a stronger adjuvant),” stated Dr. Singh. “Whether this holds in humans remains to be seen.”
Scientists are additionally testing newer vaccine platforms comparable to mRNA, which permit vaccines to be made quicker and tweaked extra simply than protein-based ones. In 2025, researchers at CureVac and the U.S. National Institute of Health (NIH) encoded the Pfs25 antigen—concentrating on the parasite’s sexual stage—into an mRNA-lipid nanoparticle. They noticed full transmission blockage in mice, with antibodies lasting over six months from simply two doses.
However, not all mRNA-based vaccine efforts are shifting forward easily. In early 2025, BioNTech’s Phase I/IIa trial for its blood-stage mRNA vaccine candidate BNT165e was positioned on medical maintain by the U.S. Food and Drug Administration (FDA). While the firm didn’t disclose the cause, it famous that discussions with regulators are ongoing. The pause highlights the hurdles of translating mRNA platforms into malaria vaccines.
“mRNA and nanoparticle platforms can certainly be explored—alone or in combination,” stated Pawan Malhotra, emeritus scientist at the International Centre for Genetic Engineering and Biotechnology (ICGEB), New Delhi. “But it’s hard to predict what will work. Plasmodium is complex, unlike bacteria or viruses.”
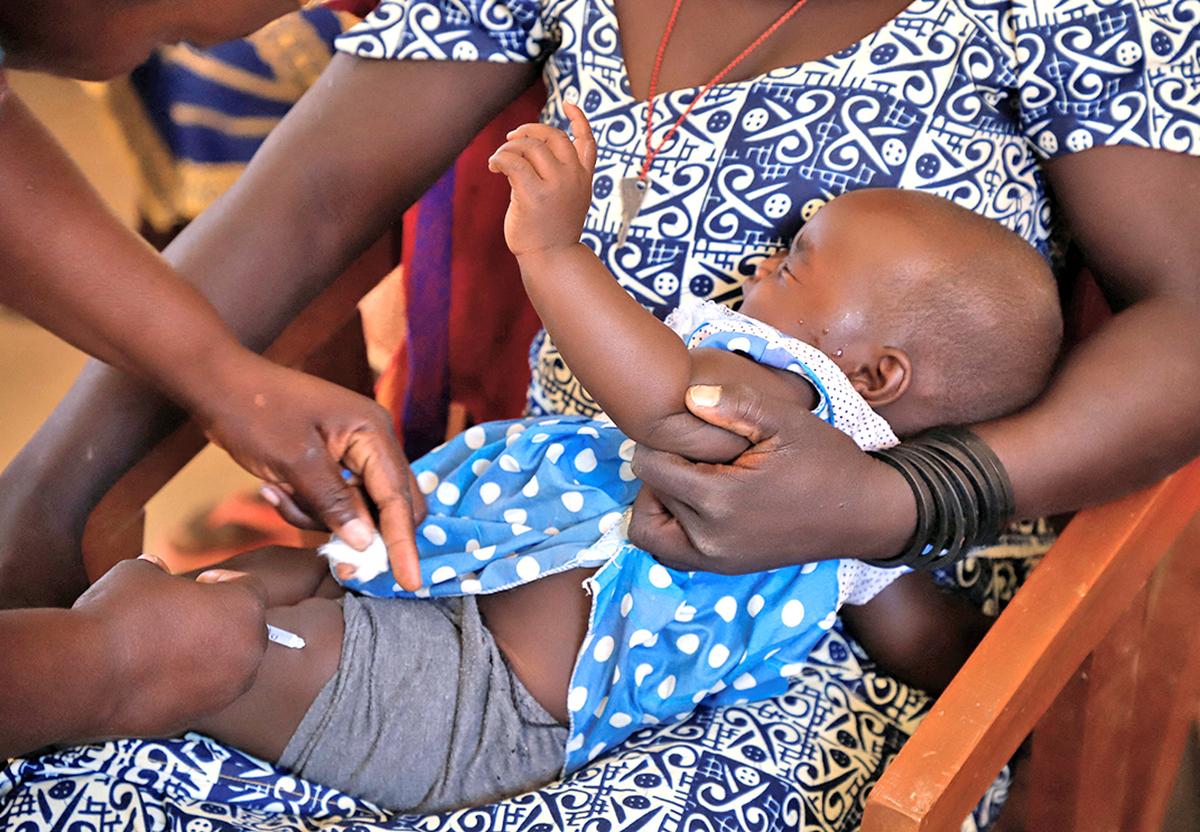
A nurse administers a malaria vaccine to an toddler at the well being heart in Datcheka, Cameroon January 22, 2024. File {photograph}
| Photo Credit:
REUTERS
Augmenting present vaccines, blocking immune evasions
Beyond boosting the energy of the immune response, scientists are additionally exploring easy methods to enhance its goal—modifying malaria antigens to assist the physique recognise the parasite extra effectively. A new experimental vaccine hyperlinks PfCSP—a floor protein from the malaria parasite—to MIP3α, a molecule that acts like a flare to attract in immune cells. In mice, it triggered stronger antibody and T cell responses than commonplace mRNA vaccines, decreasing liver-stage an infection by as much as 88%. It hasn’t but been examined in people, nevertheless it exhibits how tweaking the immune response may push malaria vaccines previous present limits.
Beyond vaccines, researchers are exploring how malaria hides from our immune system. P. falciparum makes use of RIFIN proteins to bind to immune ‘off switches’ like the LILRB1 receptor, shutting down immune cells.
It’s a tactic that helps the parasite conceal in plain sight.
A new research describes an experimental, engineered antibody, D1D2.v-IgG, designed to dam this interplay. Built from a section of the LILRB1 receptor, the antibody binds to RIFIN 110 occasions extra strongly than the pure model—outcompeting the parasite at its personal sport. By blocking this interference, it freed the physique’s LILRB1 to perform usually, restoring immune assault in lab checks. Though nonetheless untested in animals, the method may at some point help new malaria therapies or improve vaccine responses.
Gene drives and vector suppression
While engineered antibodies assault the parasite, CRISPR-based gene drives go after its vector. These instruments insert fertility-disrupting genes into mosquitoes. In a landmark research, this method worn out complete Anopheles gambiae colonies inside a 12 months—with no resistance detected.
But evolution not often performs alongside. In the wild, mosquitoes would possibly adapt, ecosystems may shift, and as soon as launched, gene drives can’t be recalled. The thought of eradicating a species raises thorny moral and ecological questions.
So, researchers are exploring subtler methods. One 2025 research edited a single letter in the FREP1 gene, blocking the malaria parasite from creating inside the mosquito. With a gene drive, this parasite-blocking trait unfold to over 90% of lab mosquitoes in ten generations—with out harming their fertility or survival. But the parasite stays below strain to evolve round the block, and contaminated mosquitoes nonetheless dwell lengthy sufficient to probably transmit malaria if the trait doesn’t saturate the inhabitants.
Another staff took a special route—engineering mosquitoes to die sooner solely when contaminated. By disabling an immune gene, they created a self-limiting suggestions loop: the extra malaria spreads, the extra it kills its personal carriers. Because this technique doesn’t assault the parasite instantly, it reduces the strain for resistance. It’s a sublime inversion—utilizing the parasite’s success in opposition to itself to shrink transmission, with out eradicating the vector or requiring good protection.
The Indian lens: challenges and the path forward
The nation goals malaria elimination by 2030. It’s an bold plan—however one which hinges on precision and persistence.
Tribal and forested districts in Chattisgarh, Jharkhand and components of the Northeast are the final strongholds of malaria—usually distant areas with restricted healthcare entry. Even in locations the place infections appear to be minimal, adults and older youngsters usually act as asymptomatic reservoirs, quietly sustaining transmission.
A municipal employee sweeps the highway in entrance of a wall portray that depicts Sir Ronald Ross, identified for demonstrating that the Anopheles mosquitoes transmit the Plasmodium parasite, offering the first evidence-based rationalization of malaria transmission
| Photo Credit:
KVS GIRI
The dual-species panorama in India additional complicates elimination efforts.
“P. cynomolgi—a monkey malaria species—is the best model for P. vivax research,” stated Dr. Malhotra. “We were developing it 20 years ago with the Central Drug Research Institute (CDRI), but strict monkey access laws and lack of scientific foresight stalled it.”
Despite these challenges in vivax analysis, efforts to develop a vaccine are gaining floor. Both Dr. Subhash Singh at ICMR and Dr. Agam P. Singh at NII affirm that P. vivax vaccine candidates are below energetic growth.
But even the most modern science wants techniques to hold it ahead.
“We need a COVID-style push,” stated Dr. Malhotra. “Industry and academia must collaborate with proper funding. We’ve developed potent therapeutic antibodies against liver-stage parasites and now need partnerships to move them forward. A lone scientist in a lab can’t do it all.” The science is advancing—nevertheless it wants infrastructure and political will to match.
Dr. Singh echoes the sentiment. “We are now concentrating on translating AdFalciVax’s promising preclinical results into trials. Successful deployment however, will require good results over multiple stages of trials as well as regulatory approvals, likely taking at least 7–8 years.” In addition, sturdy coordination between regulators, business, and researchers is required. ICMR has already floated an Expression of Interest searching for industrial companions to co-develop the vaccine. “Challenges that need to be addressed include producing GMP-grade components, developing immune biomarkers, and benchmarking efficacy against RTS,S and R21,” added Dr. Singh.
“We definitely need vaccines, antibodies, new drugs—for both P. falciparum and P. vivax,” stated Dr. Malhotra. “But that’s not enough. Doctors need training, resistance must be tracked, and vector control has to keep pace.” It have to be a full-spectrum battle—from the molecular stage to the neighborhood clinic.
India’s malaria story is not considered one of uniform burden—it’s a combat in opposition to hidden reservoirs, distant geographies, and a parasite that received’t stop. With next-gen vaccines, homegrown innovation, and rising scientific momentum, the nation stands at a essential juncture. Elimination by 2030 is not only a objective—it’s a take a look at of whether or not science, coverage, and public well being can unite to defeat an historic foe. The instruments are right here. Now, we should use them—decisively and in all places the parasite nonetheless survives.
(Anirban Mukhopadhyay is a geneticist by coaching and science communicator from Delhi. anirban.genetics@south.du.ac.in)






